Each atom will have its own
characteristic line spektrum. At
the line spectrum of hydrogen in class and saw
three lines. These red, green, and
blue lines represented emissions from different energy levels in the atom.
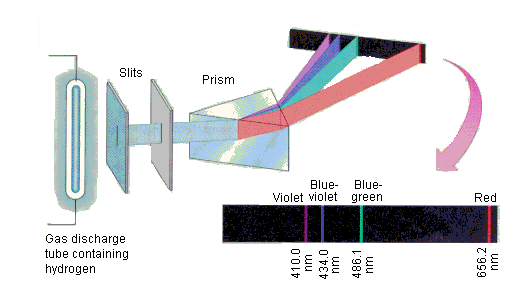
Spectrum of Hydrogen
We can look at either
absorption or emission spectra.

Using the equation for the
energy of the hydrogen levels
we can write
an equation for the change
in energy of an electron that
charges orbitals and emits or
absorbs a photon.

With this equation we can
calculate the frequency of light
emitted or absorbed when an
electron moves between orbitals of different principal
quantum numbers.

Using the equation above we can calculate
the wavelengths for various transitions
in the H-atom.

The energy required to promote an electron to n = ![]() is called the ionization
energy.
is called the ionization
energy.
(Because this is the energy
required to make an ion).
A characteristic pattern
of spectral lines, either absorption
or emission, produced by the hydrogen atom. The various series of lines are named
according to the lowest energy level
involved in the transitions that give rise to the
lines.
The Lyman series involve jumps to or from
the ground state (n=1); the Balme series (in which all the lines
are in the visible region) corresponds to n=2, the Paschen series to n=3, the Brackett series
to n=4, and the Pfund series to n=5.
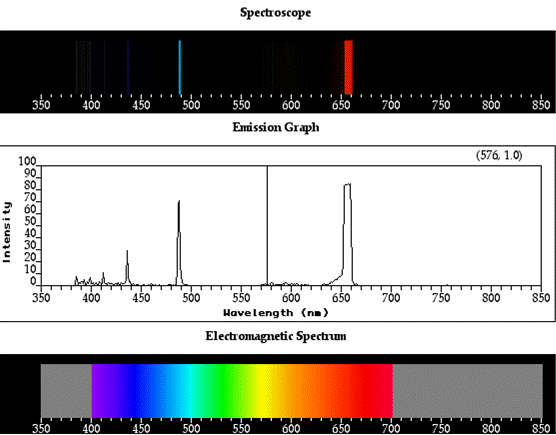
To illustrate with specific values, consider the Balmer
series, in which the four principal
lines, designated as H-alpha, H-beta, H-gamma, and H-delta, involve energy jumps of
3.02 x 10-19, 4.07 x 10-19, 4.57 x 10-19, and 4.84 x -19 joules
(J), respectively, and corresponding photons of wavelengths are 656.3, 486.1,
434.0, and 410.2 nanometers
(nm).
