Light and Other Formo f Electromagnetic Radiation
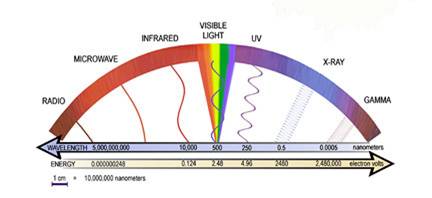
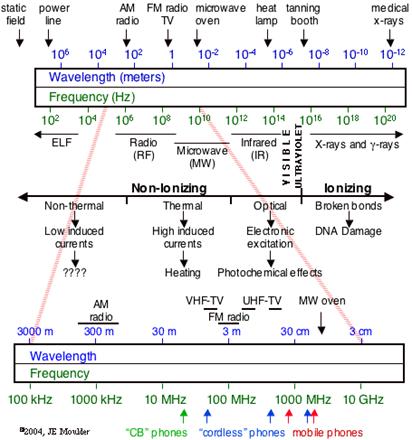
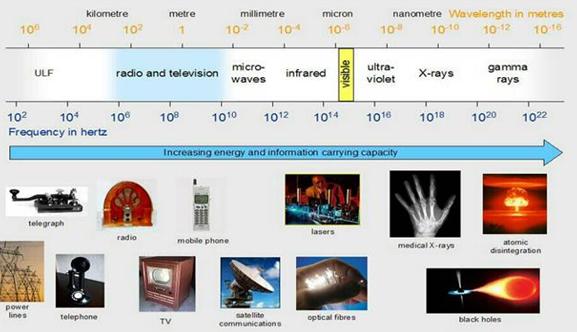
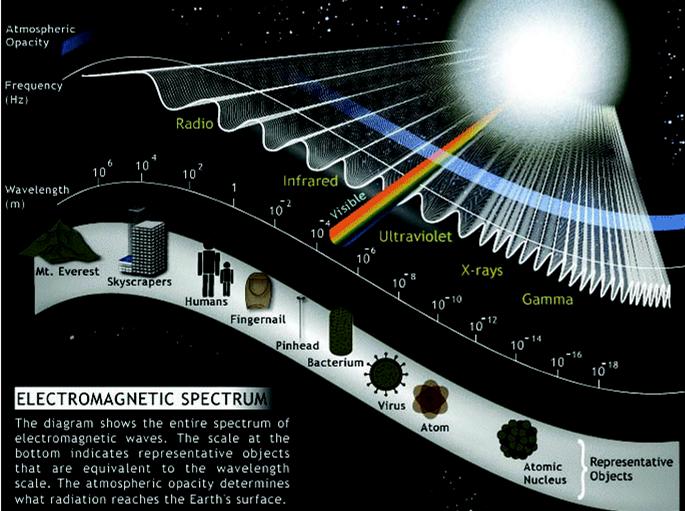
The electromagnetic spectrum includes radio and TV waves, microwaves, infrared, visible light, ultraviolet, x-rays, g-rays, and cosmic rays, as shown in the figure above. These different forms of radiation all travel at the speed of light (c). They differ, however, in their frequencies and wavelengths. The product of the frequency times the wavelength of electromagnetic radiation is always equal to the speed of light. As a result, electromagnetic radiation that has a long wavelength has a low frequency, and radiation with a high frequency has a short wavelenght
v.l = c
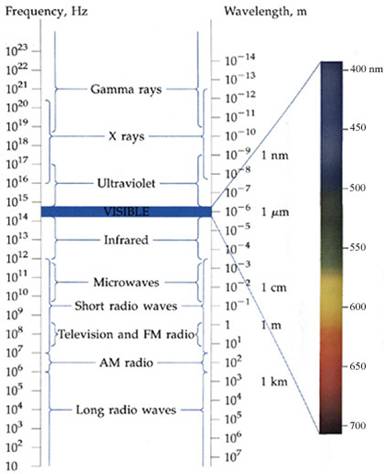
The The electromagnetic spectrum includes radio and TV waves, microwaves, infrared, visible light, ultraviolet, x-rays, g-rays, and cosmic rays, as shown in the figure above. These different forms of radiation all travel at the speed of light (c). They differ, however, in their frequencies and wavelengths. The product of the frequency times the wavelength of electromagnetic radiation is always equal to the speed of light.
Continuum, Emission, and Absorption Spectra
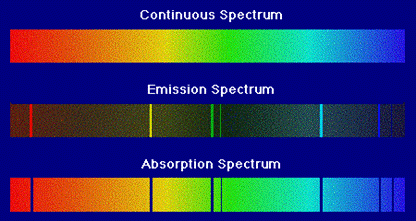
The corresponding spectrum
may exhibit a continuum, or may have
superposed on the continuum bright lines (an emission
spectrum) or dark lines (an
absorption spectrum),
as illustrated in the following figure.
Origin of Continuum, Emission, and Absorption
Spectra
The origins of these three types of
spectra are illustrated in the following figure.
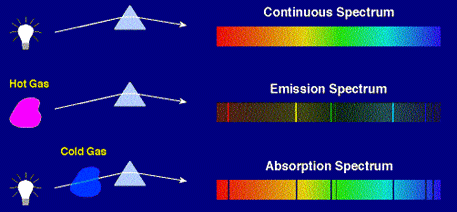
Thus, emission
spectra are produced by
thin gases in which the atoms
do not experience many collisions
(because of the low density).
The emission lines correspond to photons of discrete
energies that are emitted when excited
atomic states in the gas make
transitions back to lower-lying levels.
A continuum spectrum results when the
gas pressures are higher. Generally, solids, liquids, or dense gases
emit light at all wavelengths
when heated.
An absorption
spectrum occurs when light passes
through a cold, dilute gas and
atoms in the gas absorb at
characteristic frequencies;
since the re-emitted light is
unlikely to be emitted in the same direction as the absorbed photon,
this gives rise to dark lines
(absence of light) in the spectrum.
Application in life