Spectral Lines
We have seen how a blackbody spectrum is related to the temperature of a body. This smoothly varying spectrum is called a kontinuum spectrum because it is made up of all possible frequencies. Spectral lines, on the other hand, are narrow features superimposed on the continuum spectrum
- Spectral lines are seen when light is passed through a slit, then expanded into a spectrum. A device for doing this is called a spectroscope.
- Absorption lines are dark lines due to "missing" frequencies
- Emission lines are bright lines due to extra emission at some frequencies
- Glowing gas shows emission lines, but if you put this same gas in front of a bright source of light, the same lines appear in absorption.
- For gas of different composition, the spectral lines appear in different places, unique for each element.
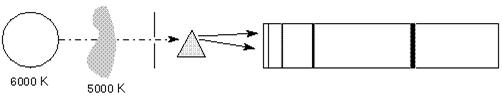
- Fig. Absorption lines
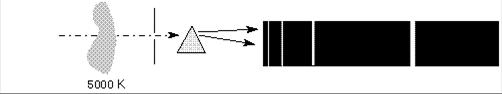
- Fig. Emission lines
SPECTRAL LINES
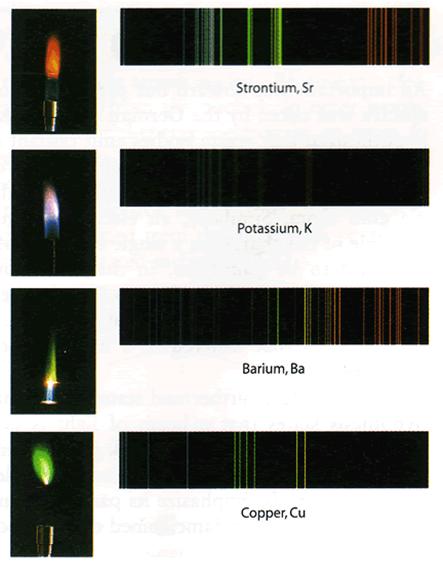
Spectral lines and spectrum
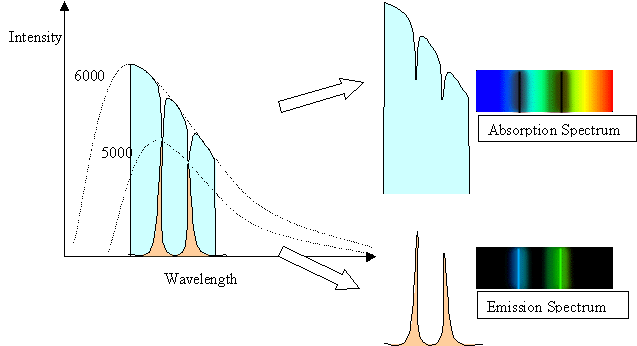
The spectral line intensities are related to the temperature of the body doing the emitting. In the case of the 5000 K gas in front of the 6000 K background, the background has a normal Planck Function blackbody spectrum except where the cooler gas is absorbing it. The depth of the lines reflect the 5000 K blackbody spectrum of the gas. In the case of the 5000 K gas with a cool background, the height or intensity of the spectral lines reflects the 5000 K blackbody curve of the gas, but only in the spectal lines. At other wavelengths, the gas has no emission, and so is dark.
.
Measuring Spectra
Spectra are measured in wavelength, either in angstrom (1 A = 10-10 m), or nanometer (1 nm = 10-9 m) units. The use of nm is more common in scientific literature, but angstroms are used often in ordinary speech. Visible light ranges from about 4000 A (blue) to 8000 A (red), or 400 nm - 800 nm.